3d Orbital Wave Function Graph. The graph on the right is a plot of values along a single line drawn through the nucleus along the z axis. 9 the probability distribution curve for 2s electron appears like that of.
Using the wavefunction it is easy to graph a variety of atomic orbitals. Orange fo r positive and blue fo r negative. Represent the wave function atomic orbital in 3d draw a 3d surface at a given value of define the surfac such that it encloses a space in which the electron spends most of its time the surface now depicts outer shape and size of the orbital.
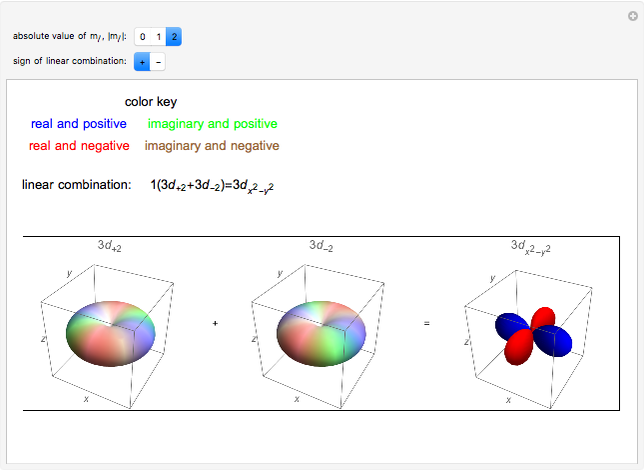
To see the elongated shape of ψ x y z 2 functions that show probability density more directly see pictures of d orbitals below.
The shapes of the first five atomic orbitals are. Each picture is domain coloring of a ψ x y z function which depend on the coordinates of one electron. However the px and py orbitals are not the shape most people are familiar with. Orange fo r positive and blue fo r negative.
